|
5/16/2023 0 Comments Does dna polyermase i fill the gap
(18) It confirmed that the enzyme was folded into two domains: an amino-terminal 8 kDa domain and the 31 kDa polymerase domain (Figure 2). (17) The structure of the unliganded apoenzyme was first reported in 1994. We next describe salient features of available crystallographic structures of the intermediates outlined in Scheme 1.Ĭontrolled proteolytic or chemical cleavage of pol β first demonstrated that it is folded into discrete domains. (16) Crystallographic structures of most of the intermediates depicted in Scheme 1 have been determined at high resolution and provide molecular insights by which an induced-fit model bestows substrate specificity (Table 1). Computational studies that bridge the observed intermediate states can be very useful in correlating structural and kinetic observations. This is especially challenging because structural models often represent static snapshots of dynamic events. In each case, the structures should be interpreted in the context of a kinetic–thermodynamic model for ligand binding and catalysis. Alternatively, reactions can be initiated in the crystal and stopped at defined moments by rapid freezing. This approach requires that intermediate states be trapped using modified enzymes, inert cofactors, or substrate analogues. Structural biology offers a unique opportunity to visualize well-populated intermediate states during catalytic cycling at a molecular level. These are not explicitly shown in Scheme 1 but are discussed in detail below. DNA synthesis also requires at least two divalent metal ions. At this point, the extended product (DNA +1) is released (single-nucleotide insertion) or serves as substrate DNA for another round of nucleotide insertion following translocation (processive DNA synthesis). 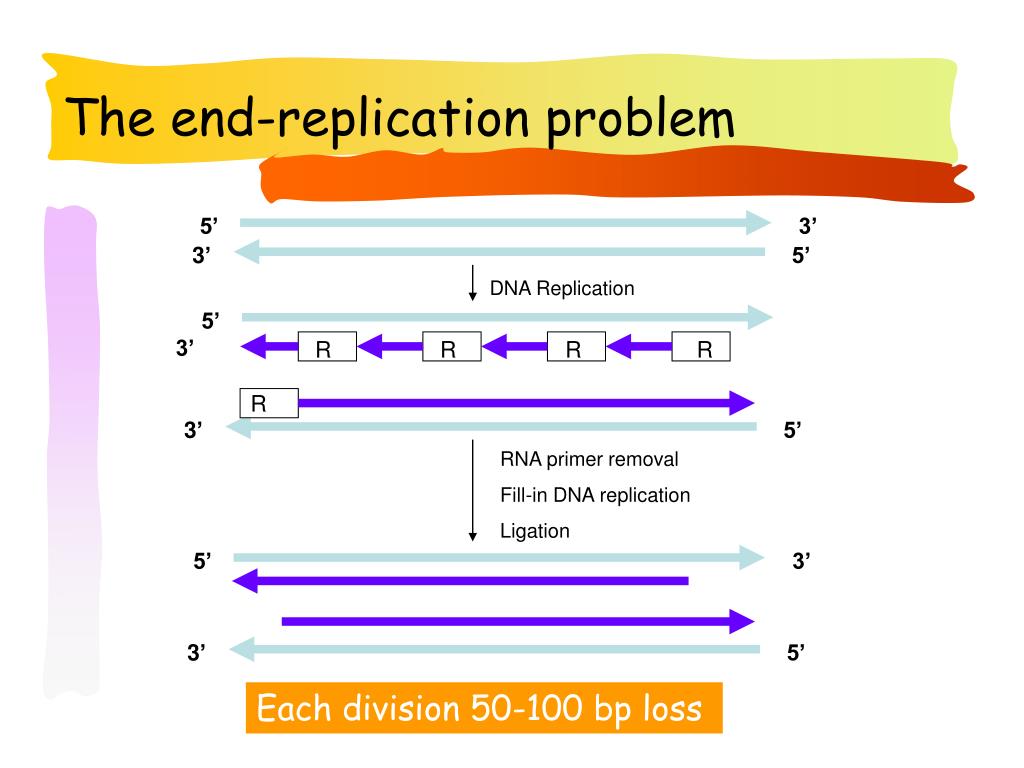
Following chemistry, the ternary product complex undergoes a conformational change (step 5) that facilitates PP i release (step 6). While many DNA polymerases undergo a large subdomain motion, more subtle conformational adjustments that either hasten or deter the subsequent chemical step (step 4) occur. The ternary substrate complex undergoes a global conformational change (step 3). (14) Steady-state kinetic analyses indicate that pol β follows an ordered binding of substrates (15) after binding DNA (Scheme 1, step 1), DNA polymerases preferentially bind a nucleoside triphosphate (dNTP) that preserves Watson–Crick hydrogen bonding as dictated by the template (coding) base (step 2). In addition, the nature (kinetic and thermodynamic) of the conformational changes(s) before and after chemistry has been the subject of a long-standing debate as well as their impact on substrate discrimination (i.e., fidelity). The detailed kinetic and equilibrium constants are sensitive to the identity of the polymerase and substrates (e.g., base and sugar of the nucleoside triphosphate and DNA sequence context). (2, 3)ĭNA polymerases are believed to follow the general pathway outlined in Scheme 1. For earlier kinetic and structural descriptions, see previous in-depth reviews. (1) This review will highlight recent advances and insights provided by the structural characterization of pol β in various liganded and conformational states. On the basis of its primary sequence, pol β belongs to the X-family of DNA polymerases. Pol β is the smallest cellular DNA polymerase (335 residues, 39 kDa) and lacks a 3′ → 5′ proofreading exonuclease activity that enhances the accuracies of replicative DNA polymerases (e.g., pol ε and pol δ). DNA polymerase (pol) β has served as a model enzyme for studying this fundamental task, providing a detailed understanding of the events during substrate selection. These enzymes are responsible for preferentially binding and incorporating a nucleotide, from a pool of chemically and structurally similar molecules, that correctly base pairs with the appropriate templating base. These structures also provide an improved framework that permits computational studies to facilitate the interpretation of detailed kinetic analyses of this model enzyme.ĭNA polymerases catalyze template-dependent DNA synthesis during genome replication and repair. Structures of these catalytic intermediates provide unexpected insights into mechanisms by which DNA polymerases enhance genome stability. In this review, recent crystallographic studies of pol β in various liganded and conformational states during the insertion of right and wrong nucleotides as well as during the bypass of damaged DNA (apurinic sites and 8-oxoguanine) are described. Pol β has been an excellent model enzyme for studying the nucleotidyl transferase reaction and substrate discrimination at a molecular level. These domains are termed the polymerase and lyase domains, respectively. Each domain contributes an enzymatic activity (DNA synthesis and deoxyribose phosphate lyase) during the repair of simple base lesions. DNA polymerase (pol) β is a small eukaryotic DNA polymerase composed of two domains.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
