|
5/17/2023 0 Comments Sequencher torrent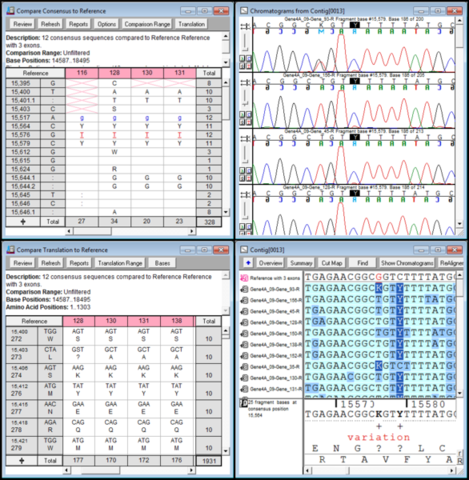
Based on the pDST results, 4% (2/50), 14% (7/50), 36% (18/50), 28% (14/50), 6% (3/50), and 12% (6/50) of cases were classified as pansusceptible TB, rifampicin-resistant TB (RR-TB), multidrug-resistant TB (MDR-TB), pre-extensively drug-resistant TB (pre-XDR-TB), extensively drug-resistant TB (XDR-TB), and others, respectively. Figure 1A and Table S1 in the supplemental material show the drug resistance patterns of the training isolates, including resistance to RIF ( n = 42 ), INH ( n = 38 ), EMB ( n = 23 ), PZA ( n = 13 ), MFX ( n = 12 ), LFX ( n = 11 ), AMK ( n = 5 ), CM ( n = 7 ), KM ( n = 6 ), SM ( n = 20 ), BDQ ( n = 10 ), CFZ ( n = 7 ), LZD ( n = 1 ), and DLM ( n = 3 ). In particular, tNGS can provide full-length sequence information with a large depth of coverage for targeted drug resistance genes, which is important for the prediction of heteroresistance ( 10).Ī training set of 50 and a challenge set of 35 well-characterized Mycobacterium tuberculosis complex (MTBC) isolates were prepared for performance assessment using multiple methods, including tNGS, WGS, Sanger sequencing, and phenotypic DST (pDST).   
With the continued expansion and reduced cost of NGS technology, WGS and tNGS could be adopted in TB control programs. Notably, whole-genome sequencing (WGS) and targeted next-generation sequencing (tNGS) have been used to predict drug resistance of TB since the WHO guideline was issued ( 9). WHO-endorsed genotypic DST (gDST) modalities, such as GenoType MTBDR plus and GenoType MTBDR sl (Bruker/Hain Lifescience, Nehren, Germany) ( 3, 4), Xpert MTB/RIF Ultra and Xpert MTB/XDR (Cepheid, Sunnyvale, CA, USA) ( 5, 6), and Truenat MTB-RIF-Dx (Molbio Diagnostics, Goa, India) ( 7), have limited detection capabilities, especially when rare or novel mutations are located outside detection regions of drug resistance-associated genes ( 8). Rapid, accurate, and comprehensive diagnosis of DR-TB is crucial to provide personalized and precision medications for better treatment outcomes. It usually takes months to obtain conventional DST results, which impacts patient outcomes and poses great risks to public health and challenges to global TB control. The current standard for comprehensive DR-TB diagnosis is culture-based DST. Our customized novel whole-gene-based tNGS panel is highly consistent with pDST and WGS for comprehensive and accurate prediction of drug resistance in a strengthened and streamlined DR-TB laboratory program. The agreement between tNGS and pDST was almost perfect for RIF, INH, EMB, MFX, LFX, AMK, CM, KM, SM, BDQ, and LZD (kappa value = 0.807 to 1.000) and substantial for PZA (kappa value = 0.791). Regarding the limit of detection, the tNGS assay detected 2.9 to 3.8% minority variants in 4% mutant mixtures. Phenotypic drug susceptibility testing (pDST) results were used as gold standards. Comparative variant analyses of the DR genes were performed using Sanger sequencing and whole-genome sequencing (WGS). We selected 50 and 35 Mycobacterium tuberculosis isolates with various DR profiles as the training set and the challenge set, respectively. To provide comprehensive drug susceptibility information and to address mutations missed by available commercial molecular diagnostics, we developed and evaluated a tNGS panel with 22 whole-gene targets using the Ion Torrent platform to predict drug resistance to 14 drugs, namely, rifampicin (RIF), isoniazid (INH), ethambutol (EMB), pyrazinamide (PZA), moxifloxacin (MFX), levofloxacin (LFX), amikacin (AMK), capreomycin (CM), kanamycin (KM), streptomycin (SM), bedaquiline (BDQ), clofazimine (CFZ), linezolid (LZD), and delamanid (DLM). Targeted next-generation sequencing (tNGS) has emerged as an alternative method for detecting drug-resistant tuberculosis (DR-TB).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
